 How many electrons are in their outer shell? (Hint: it' s the same as their oxidation (charge) number or group number.) two s electrons 16. Do these metals occur freely in nature? the second most reactive family of elements. Go back and click on Alkaline Earth Metals (left bar) and answer these questions. What happens when they are exposed to water? produce heat, hydrogen gas, and the corresponding metal hydroxide. 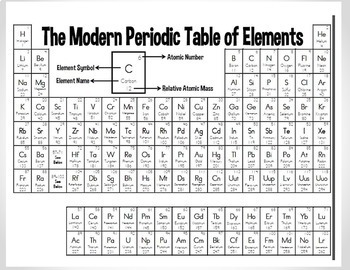
Name the two most reactive elements in this group? Francium the most reactive, followed by cesium and g. What are the three characteristics of ALL metals? - good conductors of heat and electricity - malleable - ductile e. How many electrons are in their outer shell? one electron d. Do these metals occur freely in nature? not found in free state c. are these metals reactive? Alkali metals are among the most reactive metals b. What is the group number? represent the valence electron (s) present in the elements belonging to a specific group. Click on Alkali Metals (left side menu bar) and answer the following questions. Families of the Periodic Table Website #3: chemicalelements/ 14. NUMBER THE GROUPS AND PERIODS ON YOUR PERSONAL PERIODIC TABLE. It is very stable with only two electrons in its outer orbital (valence shell). Helium: is different from all of the other elements. Why do Hydrogen and Helium appear to be ‘separated’ from the rest of the table? because it has a filled valence electron shell Hydrogen: in its neutral form, does not have a neutron. Every element in group one has one electron in its outer shell. Elements in the same group share the same number of what? valence electrons. Groups are: a column in the periodic table of the chemical elements Periods are: A period is a horizontal row of the periodic table. Why are the elements placed in specific places on the Periodic Table? because of its atomic structure. Atomic # Website #2: chem4kids/elements/table.html atomic Label EACH part of the block from the periodic table at right. What can you do to find the mass number? mass number = protons + neutrons. You can’t get the mass number from the periodic table. How’s the atomic weight/mass found? adding the total number of protons and the total number of neutrons of that particular isotope. How would you determine the number of neutrons in an atom? Show as a formula! equal to the difference between the mass number of the atom (M) and the atomic number (Z). What does the atomic weight of an element represent? The total weight of an atom If an atom is neutral, then the number of electrons is equal to the number of: the number of protons What does the atomic number of an element represent? the number of protons in the nucleus of an atom Review Website #1: education.jlab/qa/pen_number.html Information and obtain the answers from the websites provided. To answer the questions, READ the page of Not to miss out, element 102, Nobelium, No, is named in honor of Alfred Nobel, who set aside his vast fortune to establish Nobel Prizes.# Getting to know the Periodic Tableĭirections: Go to the websites that are linked for each section. Mendeleev never received a Nobel Prize for his work, but element 101 was named Mendelevium, Md, after him. For instance they discovered phosphorus when they isolated it from urine. Over time these gaps have gradually been filled in as scientists unearthed new elements. Not only did Mendeleev arrange the elements in the correct way, but he also had the foresight to leave gaps for undiscovered elements. At that time, he had only 50 elements to arrange. He wrote the properties of the elements on pieces of card and rearranged them until he realised that, by putting them in order of increasing atomic weight, certain properties of elements regularly occurred. Then in 1869, a Russian scientist called Dmitri Mendeleev produced one of the first practical periodic tables.  
Several other attempts were made to group elements together over the coming decades. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths. The discovery of other elements followed regularly and soon it became necessary to arrange them in some sort of order. However, the first scientific discovery of an element occurred in 1649 when Hennig Brand discovered phosphorous. Copper has been used by humans for as much as 7000 years and elements such as gold, silver, tin, lead and mercury have been known for many thousands of years. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
